|
Compared to calcium which only has its 4s electrons as mobile charge carriers, transition metals also have the 3d electrons as mobile charge carriers. The delocalized electrons of metals serve as mobile charge carriers for the conduction of electricity. It is shown in the table below that, calcium has a lower first ionisation energy compared to the first row transition metal and most of the transition metals except Hafnium and Tantalum. Generally, transition metals have a smaller ionic radius as compared to calcium.Īcross a period, the first ionisation energy increase with increasing atomic number as the positive charge of nucleus increase, forces of attraction between the nucleus and valence electron strengthen, and so more energy is required to give out that electron. Ca 2+ has an ionic radius of 114 pm while Fe 2+ has an ionic radius of 77 pm. Comparing Ca 2+ with Fe 2+, in this case the degree of ionisation is the same so the two can be contrasted. Radii decrease due to the face that across the period there is more proton, and when there is a lost of electrons, there will be a stronger electrostatic attraction between the nucleus and the outermost shell. Across a period, radii of ions decrease until we reach the negatively charged ions. Generally, transition metals have a smaller atomic radii compared to calcium.ĭown the group, extra layers of electrons are added leading to ions getting bigger. Therefore, atomic radius is smaller for the iron. Compare calcium with iron, they have same shielding shells, but iron has a stronger force of attraction of the valence electrons due to a greater amount of positive charged proton. As density is defined as mass per unit volume, if there is a same volume of calcium and iron were used to measure density, by having a greater mass, the iron will result in greater density.Īcross the period, the number of shielding shells remains the same, but positive charge of nucleus increase, leading to a stronger force of attraction between the nucleus and the valence electrons, resulting in a smaller atomic radius. Compared to calcium, transition metals such as iron and copper has a greater mass. Compared to calcium which only has the 2s electrons to be delocalized.Īcross the period, masses of elements increase. As seen in transition metals, not only some 3d electrons are delocalized, but also the 4s electrons. Element (sym),, Mass, density, oxid s, mp, bp. 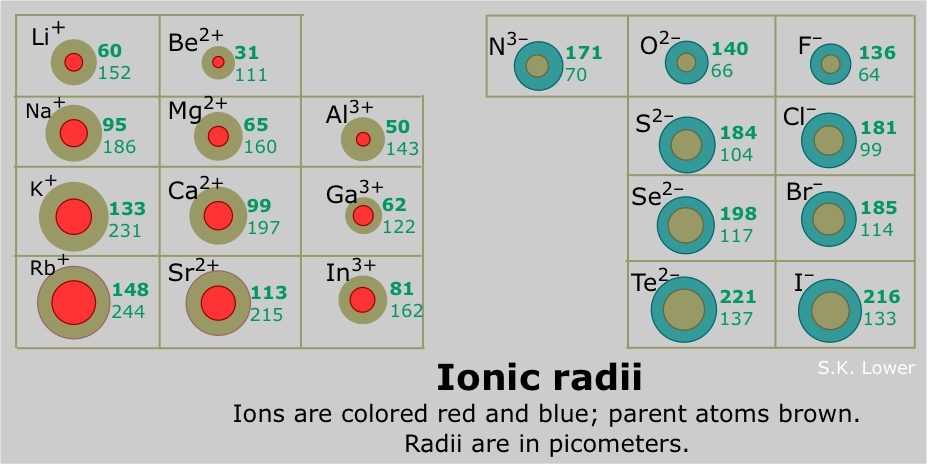
More delocalized electrons, the stronger the metallic bonds. Metallic bonding is defined by delocalization of valence electrons, leaving the metal cations to be embedded in a sea of delocalized electrons. 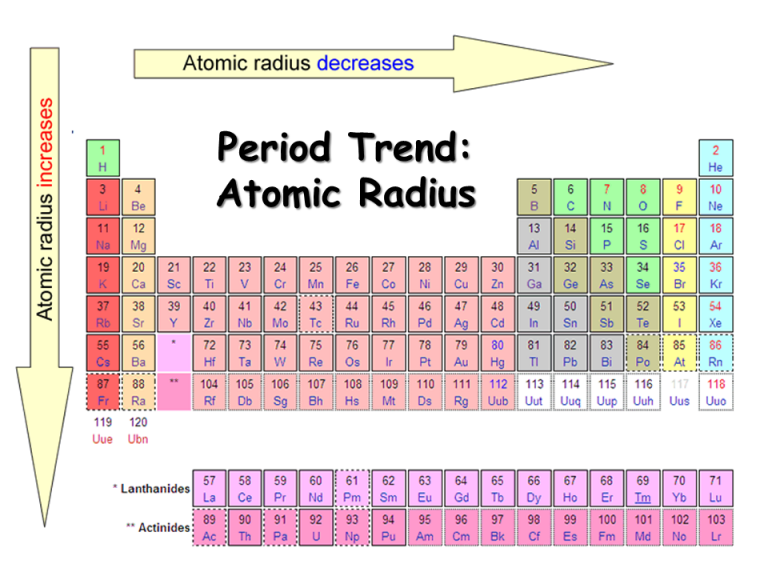
Transition metals have high melting points due to their strong metallic bonding.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
